 
However, this equation isn't balanced because the number of atoms for each element is not the same on both sides of the equation. For example, in the reaction of hydrogen (H₂) with oxygen (O₂) to form water (H₂O), the chemical equation is: It shows the reactants (substances that start a reaction) and products (substances formed by the reaction). calcium hydroxide + carbon dioxide = calcium carbonate + waterĮxamples of the chemical equations reagents (a complete equation will be suggested):Ī chemical equation represents a chemical reaction.PhCH 3 + KMnO 4 + H 2SO 4 = PhCOOH + K 2SO 4 + MnSO 4 + H 2O.To enter an electron into a chemical equation use + H 2O.Compare: Co - cobalt and CO - carbon monoxide Always use the upper case for the first character in the element name and the lower case for the second character.Enter an equation of a chemical reaction and click 'Balance'.Instructions on balancing chemical equations: Please tell about this free chemistry software to your friends! Now we substitute the variables in the original equations with the values obtained by solving the linear algebra system and arrive at the fully balanced equation: To get to integer coefficients we multiply all variable by 2 Solving this linear algebra system we arrive at: Now we assign a=1 and solve the system of linear algebra equations: Now we write down algebraic equations to balance of each atom: Let's balance this equation using the algebraic method.įirst, we set all coefficients to variables a, b, c, d. 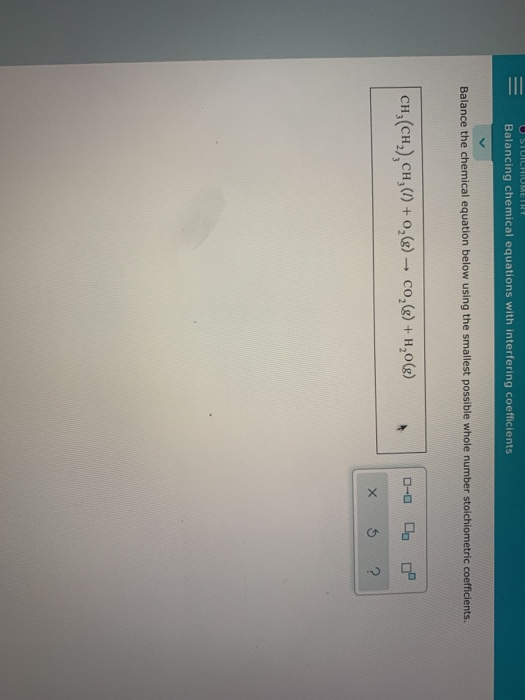
H is not balanced: 2 atoms in reagents and 6 atoms in products.Īll atoms are now balanced and the whole equation is fully balanced:īalancing step by step using the algebraic method Let's balance this equation using the inspection method.įor each element, we check if the number of atoms is balanced on both sides of the equation.Ĭ is not balanced: 1 atom in reagents and 2 atoms in products. Balancing step by step using the inspection method
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
